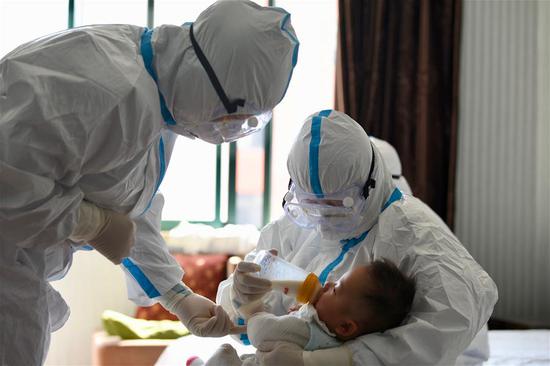
A rapidly developed coronavirus vaccine will be tested on healthy volunteers in the U.S. in April, the director of the National Institute of Allergy and Infectious Diseases (NIAID) said Tuesday.
The vaccine, by drugmaker Moderna and NIAID, will be tested on 20 to 25 humans to determine whether two doses will be safe and generate an immune response to protect against infection, with results available in July or August, NIAID's Dr Anthony Fauci told The Wall Street Journal.
NIAID is part of the National Institutes of Health (NIH), an agency of the U.S. Department of Health and Human Services. The Coalition for Epidemic Preparedness Innovations (CEPI), an international funding body, is also backing the effort.
Also on Tuesday, NIH announced that the first clinical trial testing of Gilead Sciences' antiviral drug remdesivir in a hospitalized patient with the coronavirus has started, according to Reuters.
The participant is an American who was repatriated after being quarantined in Japan aboard the cruise ship Diamond Princess and is being conducted at the University of Nebraska Medical Center in Omaha, NIH said.
The World Health Organization (WHO) said Monday that remdesivir is showing signs of helping to treat the coronavirus.
A successful trial of Moderna's vaccine — mRNA-1273 — would be followed by further studies and regulatory hurdles, Fauci said, meaning a coronavirus vaccine would not be ready for widespread distribution until at least next year.
Moderna said the vaccine was developed after obtaining genetic information on the coronavirus from China.
Referring to the speed of the drug's development and expected clinical trial, Fauci said it is "unquestionably the world indoor record. Nothing has ever gone that fast."
However, it's uncertain whether the drug will work, considering that the gene-based technology it uses hasn't yet resulted in an approved human vaccine.
Even if it isn't ready for widespread distribution until next year, Fauci told the newspaper that it's possible the spread of the coronavirus could lessen during warmer months but return next winter and become a seasonal virus like the flu.
"The only way you can completely suppress an emerging infectious disease is with a vaccine," he said. "If you want to really get it quickly, you're using technologies that are not as time-honored as the standard — what I call antiquated — way of doing it."
Moderna was founded in 2010 to develop drugs and vaccines. In 2018 it opened a manufacturing site in Norwood, Massachusetts, near the company's Cambridge headquarters. The company is targeting cancer, heart disease and infectious diseases. It hasn't brought any drugs or vaccines to market.
The first trial will be conducted at NIAID's clinical trials unit in Bethesda, Maryland, where the agency is based. If the first one is successful, a second trial of hundreds or thousands of participants could begin, which could take six to eight months, he said.
That trial could be conducted partly in the U.S. but also in other regions where the virus is spreading, so the testing could gauge whether the vaccine reduces infection rates.
If the second trial is positive, the vaccine could be ready for widespread use, Fauci said. How widely the virus has spread by then will determine whether it is given to targeted groups, such as healthcare workers, or more broadly to the general population, he said.
Health officials and pharmaceutical companies around the world are racing to identify treatments or a vaccine to help fight the coronavirus, which has infected more than 80,000 people around the world.
Johnson &Johnson, GlaxoSmithKline and Gilead are among the drugmakers working on vaccines, as are government scientists, including some at NIAID.