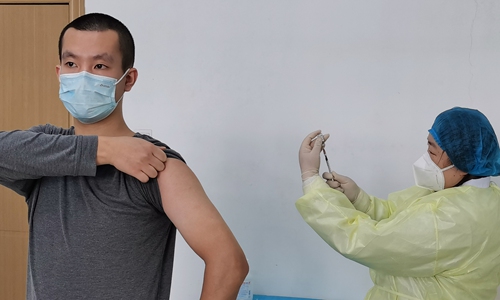
Zhu Aobing takes his vaccine injection on March 19 in Wuhan. (Photo/Courtesy of Zhu Aobing)
'Further procedures' to go before roll-out, emergency use by year-end
China's first group of volunteers in Phase I clinical trials for a COVID-19 vaccine expressed confidence in its success, as researchers work tirelessly on a vaccine to combat the deadly virus which has so far killed more than 12,000 people across the world.
The volunteers, all Wuhan residents between 18 and 60, were injected on Friday, which experts said marked a significant breakthrough that highlights China's strong research capabilities.
The vaccine was developed by Tianjin-based biotechnology company CanSino Biologics Inc and a research team headed by Chen Wei, an expert in biological hazard prevention and control from the People's Liberation Army (PLA) Academy of Military Medical Sciences and major general of PLA.
The research teams appealed for volunteers for the Phase I trial on March 17 after gaining approval on March 16, 19 hours after the U.S. announced its human trials. There are 108 volunteers divided into three groups, who received different doses of the shots.
One of the volunteers is Zhu Aobing, 28, who served in the military. He received his first dose on Thursday.
Having witnessed the death of his friend's mother from the novel coronavirus, he said he wanted to find a way to help his hometown Wuhan out of its plight.
"When I heard the good news that our self-developed vaccine was going into clinical trials, it was like a light shining into the gray city. I hope to witness the vaccine succeeding and benefiting everyone. I had no hesitation in signing up," Zhu told the Global Times on Sunday.
He said the volunteers were carefully selected, and had to undergo blood draws and physical examinations, including a CT scan, nucleic acid test, throat swab, anal swab, blood pressure check, pulse and body mass index. Past medical history and blood transfusion history were also recorded.
"I heard that more than 3,000 volunteers signed up, but only about 100 got the vaccine," said Chen Mengjiao, another volunteer from Wuhan.
Volunteers had to sign a letter of consent and informed of all possible risks. The rigorous procedure and detailed instructions gave Zhu confidence.
"After the injection, each person's temperature is monitored 24/7 by a device attached to the armpit with adhesive tape so the researchers have continuous real-time data," Chen said.
With absolute trust in the professionalism of Chinese researchers and the strong credibility of the PLA scientific team, Chen Kai, 36, signed up to be a volunteer immediately.
Expert teams will closely observe volunteers on the day of injection and post-injection, will visit participants on day 3, day 7, day 10, day 14, day 28, in month 3 and month 6, Chen said.
Most of the first volunteers are males above 30 years old, while there is a need for female volunteers under 23 or over 55, Chen said.
Some volunteers experienced the normal side effects of vaccines, like soreness and a temporary increase in temperature, Zhu said, but he has not experienced any adverse effects.
Volunteers are provided with special nutritious meals every day and are not allowed to smoke, drink or go out without permission.
They are quarantined in a single room at a nursing home for 14 days, and will be released when antibodies against the virus are detected. They will be monitored for six months for adverse reactions and will have regular blood samples taken.
Except being paid 800 yuan ($112.74) for each blood draw, the volunteers joined the trial on a voluntary basis with no extra reimbursements.
Netizens applauded these guinea pigs who are rendering Chinese society an extraordinary service to test the safety and efficacy of the vaccine while exposing themselves to potential risks.
More than 20 vaccines for COVID-19 are under development according to World Health Organization. Countries with strong medical research and development capability like Germany and Japan have both announced vaccine projects, but the U.S. and China are the only two that have entered clinical trials.
U.S. researchers administered the first injections in the country's first trial of an experimental coronavirus vaccine on March 16.
It will take at least one year to complete further procedures before the vaccine can be rolled out, according to a Beijing-based immunology expert who asked to remain anonymous.
But some products may be available for emergency use by the end of this year, said the expert.